Lipid Removal for Phenotypic Cell Response in Cancer Research
Background
Biotech Support Group (BSG) is a leading provider of sample preparation products. We are dedicated to create new
methods and applications to drive efficient workflows and better data quality for all proteomic and biomarker
analyses. Of special importance is the value created when certain families of biomolecules can be evaluated with
respect to cell response and viability. Lipids in particular have been associated with phenotypic changes in cancer
cell lines.
The Challenge: Removing lipids without introducing artifacts that affect cell
viability
Many lipid removal methods utilize solvents, Freon or chloroform. Some methods use solid-phases that suffer from
non-specific protein binding.
The Solution: Cleanascite™ Lipid Removal Reagent
Cleanascite™ is the only suspension reagent that eliminates these artifacts, making its selectivity profile for
lipids un-rivaled in the bio-research products industry. As a result, it is ideal to clear lipid-associated matrix
effects, to determine their influence on cell response assays.

The Outcomes
Demonstrating its use for a variety of cancer research, published reports describe the use of Cleanascite™ to
characterize the influence of lipids and other factors bound to lipids, on cell response in cancer.
Tumor-associated Macrophages – Two reports
First Report
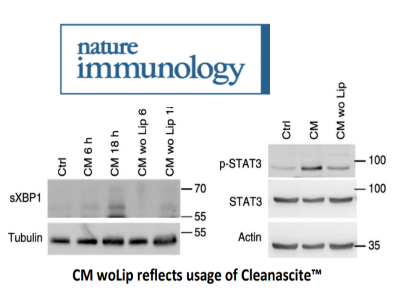
As part of the study, a comparison of culture media (CM) with and without lipids (woLip) was made. The article
states “To generate culture media without lipids, …CM was treated with Cleanascite according to the manufacturer’s
instructions.”. The authors uncover the unexpected roles of tumor-cell-produced lipids that simultaneously
orchestrate macrophage polarization and survival in tumors via induction of ER stress responses and reveal
therapeutic targets for sustaining host antitumor immunity.
Learn More
Second Report
Tumor-infiltrating monocytes can mature into Macrophages that support tumor survival or antitumor properties. To
explore mechanisms steering Macrophage maturation, the authors assessed the effects of supernatants from squamous
cell carcinoma cell lines (FaDu and SCC) on monocyte-derived Macrophage maturation. To assess depletion of fatty
acids from tumor supernatants, tumor-conditioned medium was treated with Cleanascite, stating “Depletion of Fatty
acids with Cleanascite from FaDu or SCC supernatants largely reversed the phenotypic changes in Macrophages
otherwise observed by incubating monocytes in these supernatants”.
Learn More
Omental conditioned media (OCM) – 4 separate reports
First report
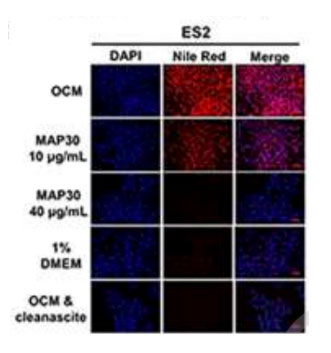
The investigators report that a bioactive protein, MAP30, isolated from bitter melon seeds exhibited potent
anticancer and anti-chemoresistant effects on ovarian cancer cells. To support the analysis of MAP30 in ovarian
cancer cells, the authors state “Nile Red fluorescence of lipid-loaded ES2 (human epithelial ovarian cancer cells)
in OCM culture for 48 h. …OCM (Omental conditioned medium) with Cleanascite™ treatment were used as negative
controls.”.
Learn
More
Second report
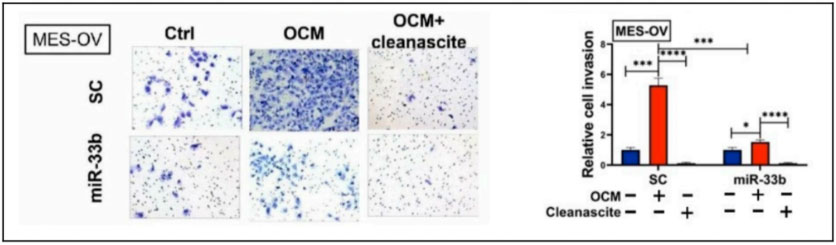
To examine whether fatty acids in OCM are the main source of energy for tumors, all fatty acids in OCM were first
removed by Cleanascite™ Lipid Removal Reagent. XTT cell viability analysis was performed and showed that the cell
growth
rate of ES-2 and MES-OV cells was remarkably reduced when cocultured in lipid-depleted OCM and “both miR-33b
overexpression and depletion of fatty acids by Cleanascite in OCM significantly impaired ovarian cancer cell
migration
and invasion.”
Learn More
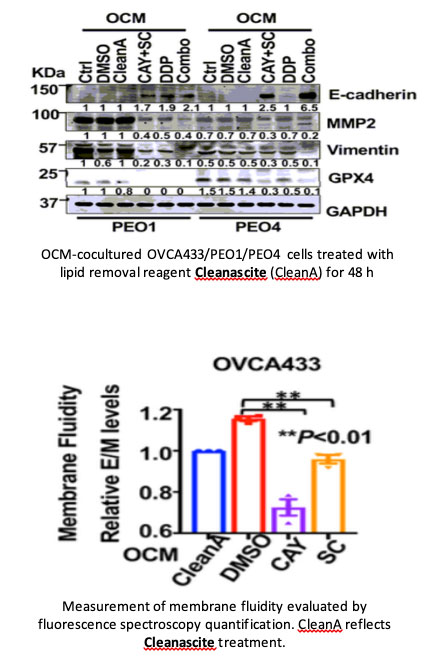
Third Report
In this study, the authors investigated whether reprogramming of lipid metabolism in ovarian cancer cells could
modulate cell viability and aggressiveness. The article states: “To determine whether fatty acids in OCM are the
primary energy source, fatty acids from OCM was first removed by Cleanascite™ Lipid Removal Reagent... Then, XTT
cell proliferation assays showed that the growth rate of ovarian cancer cells was remarkably reduced in cells
cultured in Cleanascite™-treated OCM (Fig. 2d). Likewise, co-treatment with Cleanascite™ and OCM significantly
attenuated the increased cell migration and invasion capacities of ES-2 and SKOV3 cells. These findings suggest that
the fatty acid-enriched OCM provides as an energy source for supporting tumor growth and aggressiveness of ovarian
cancer cells.”. The authors conclude that targeting the lipid metabolism signaling axis impedes ovarian cancer
peritoneal metastases.
Learn More
Fourth Report
Malignant ascites in peritoneal metastases is a lipid-enriched microenvironment and is frequently involved in the
poor prognosis of epithelial ovarian cancer (EOC). However, the detailed mechanisms underlying ovarian cancer (OvCa)
cells dictating their lipid metabolic activities in promoting tumor progression remain elusive.
Compared with the negative controls (OCM pretreated with the lipid removal reagent, Cleanascite), OvCa cells
cocultured in the lipid-enriched OCM showed an increase of 18% in membrane fluidity.
Learn More

Methylmalonic acid (MMA) complexed with lipidic structures
The authors describe how metabolic alterations that occur with age can produce a systemic environment that favors the
progression of tumors. Specifically, that methylmalonic acid (MMA), a by-product of propionate metabolism, is
upregulated in the serum of older people and functions as a mediator of tumor progression. To support this analysis,
the authors state “HS (human serum) samples were manipulated to assess the components … that might facilitate
entrance of MMA into cells. To delipidate the HS, Cleanascite Lipid Removal Reagent (Biotech Support Group) was
used…” This resulted in a reduction in total serum MMA levels and was sufficient to abrogate the pro-aggressive
phenotype. The data show that MMA, complexed with lipidic structures, is a circulatory factor that contributes to
the pro-aggressive effects of ageing in cancer cells and is sufficient to drive tumour progression and
aggressiveness.
Learn
More
Intratumoral Treg cells
Depleting regulatory T cells (Treg cells) to counteract immunosuppressive features of the tumor microenvironment
(TME) is an attractive strategy for cancer treatment. However, systemic impairment of their suppressive function
limits its therapeutic potential. The use of Cleanascite™ helped demonstrate that intratumoral Treg cells increase
lipid metabolism and CD36 expression. The study concludes that CD36 targeting elicited additive antitumor responses
with anti-programmed cell death protein 1 therapy. The findings uncover the unexplored metabolic adaptation that
orchestrates the survival and functions of intratumoral Treg cells, and the therapeutic potential of targeting this
pathway for reprogramming the tumor microenvironment.
Learn More
To download whitepaper entitled “Cleanascite™ - Lipid Removal and Cell Response Applications”, visit:
https://www.biotechsupportgroup.com/v/vspfiles/templates/257/pdf/CleanasciteCellResponseReferenceApplications113021.pdf
For more information visit: Cleanascite™ Lipid Removal Reagent and Clarification, at
http://www.biotechsupportgroup.com/Cleanascite-Lipid-Removal-Reagent-p/x2555.htm
|